Research
DYNAMIC SELF-ORGANIZATION OF LIVING MATTER ON MULTIPLE LENGTH SCALES
NOISE IN BIOLOGICAL SYSTEMS

Understanding the role of noise in crowded biological systems far from equilibrium is still in its infancy due to the systems’ enormous complexity. In fact, even identifying and quantifying the type and correlations of system-inherent noise in biological reaction networks is a persistent problem. We are employing advanced light microscopy methods and various simulation approaches to investigate, for example, complex intracellular transport processes, i.e. the heterogeneous, often anomalous, and partially anisotropic motion of macromolecules in cellular fluids and membranes. With this combined approach we aim to uncover consequences of crowding and noise on intracellular biochemical reactions, protein sorting, and intracellular pattern formation.
ORGANELLE FORMATION AND HOMEOSTASIS
Organelles in eukaryotic cells often have complex shapes that deviate significantly from simple spheres. A prime example is the endoplasmic reticulum (ER) that forms an extensive network of membrane tubules in many mammalian cell types and in reconstitution assays in vitro. Mitochondrial networks, emerging due to elaborate fusion-fission cycles of mitochondrial particles, are another prominent example. Despite the successful hunt for molecular factors that are responsible for the making, breaking, and shaping of these organelles, a comprehensive understanding of their dynamic morphogenesis is still missing. We are using advanced light microscopy techniques in conjunction with simulation approaches to gain a deeper understanding on the dynamic self-assembly and maintenance of ER and mitochondria in mammalian cells.
SELF-ORGANIZATION OF CELLS DURING EMBRYOGENESIS
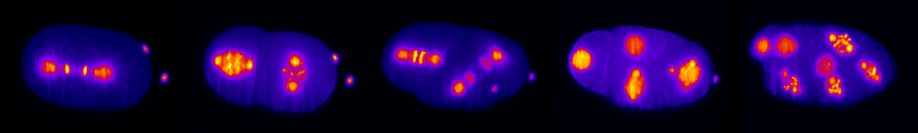
Biochemical signaling pathways in developmental processes have been extensively studied, yet the role of physical forces and mechanical cues during embryogenesis have been explored in much less detail. To address the interplay between mechanical and biochemical cues, we are mainly utilizing the developmental model organism Caenorhabditis elegans and selective plane illumination microscopy. We wish to elucidate, for example, how the embryo organizes the sequence of cell divisions, how cells determine where they need to go during development, and how these migration patterns feed back into the cell, e.g. via cell polarization.
